SPRAVATO®▽ (Esketamine) Nasal Spray Approved in Europe for Adults with Treatment-Resistant Major Depressive Disorder | Business Wire

J&J's ketamine depression drug Spravato spurned by England's influential cost watchdogs | Fierce Pharma
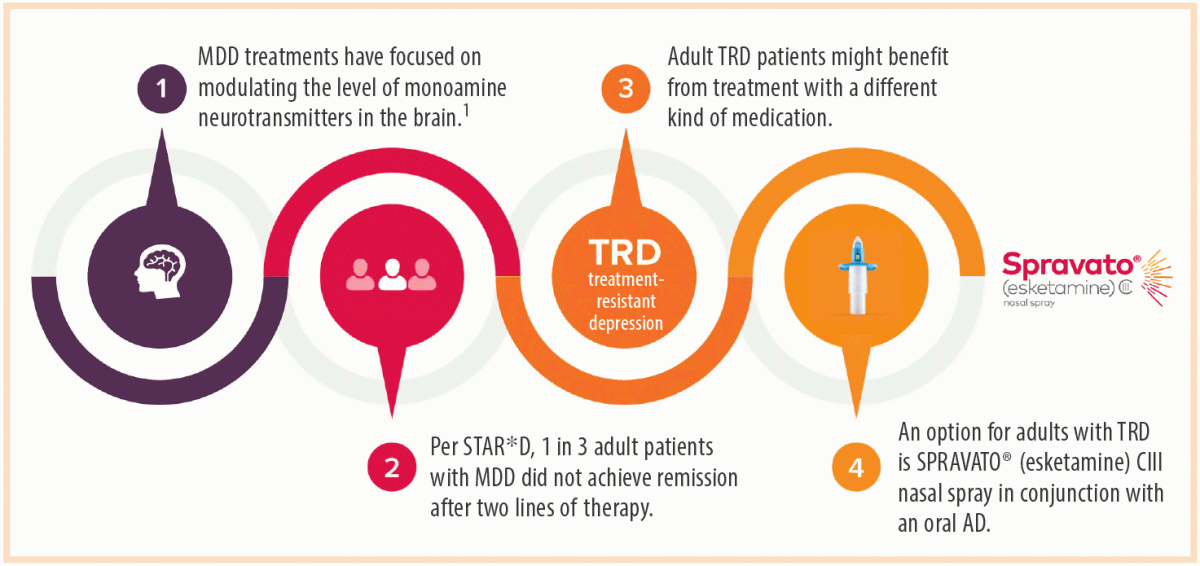
SPRAVATO® (esketamine) CIII Nasal Spray: The First and Only N-Methyl D-Aspartate (NMDA) Receptor Antagonist Approved in Conjunction With an Oral Antidepressant for the Treatment of Adults With Treatment-Resistant Depression | Psychiatrist.com

New Spravato FDA Approval Opens The Door to New Patients, Says Sandhya Prashad, M.D. - Sandhya Prashad, MD
FDA approves esketamine nasal spray, the first new major depression drug in more than 30 years | TechCrunch

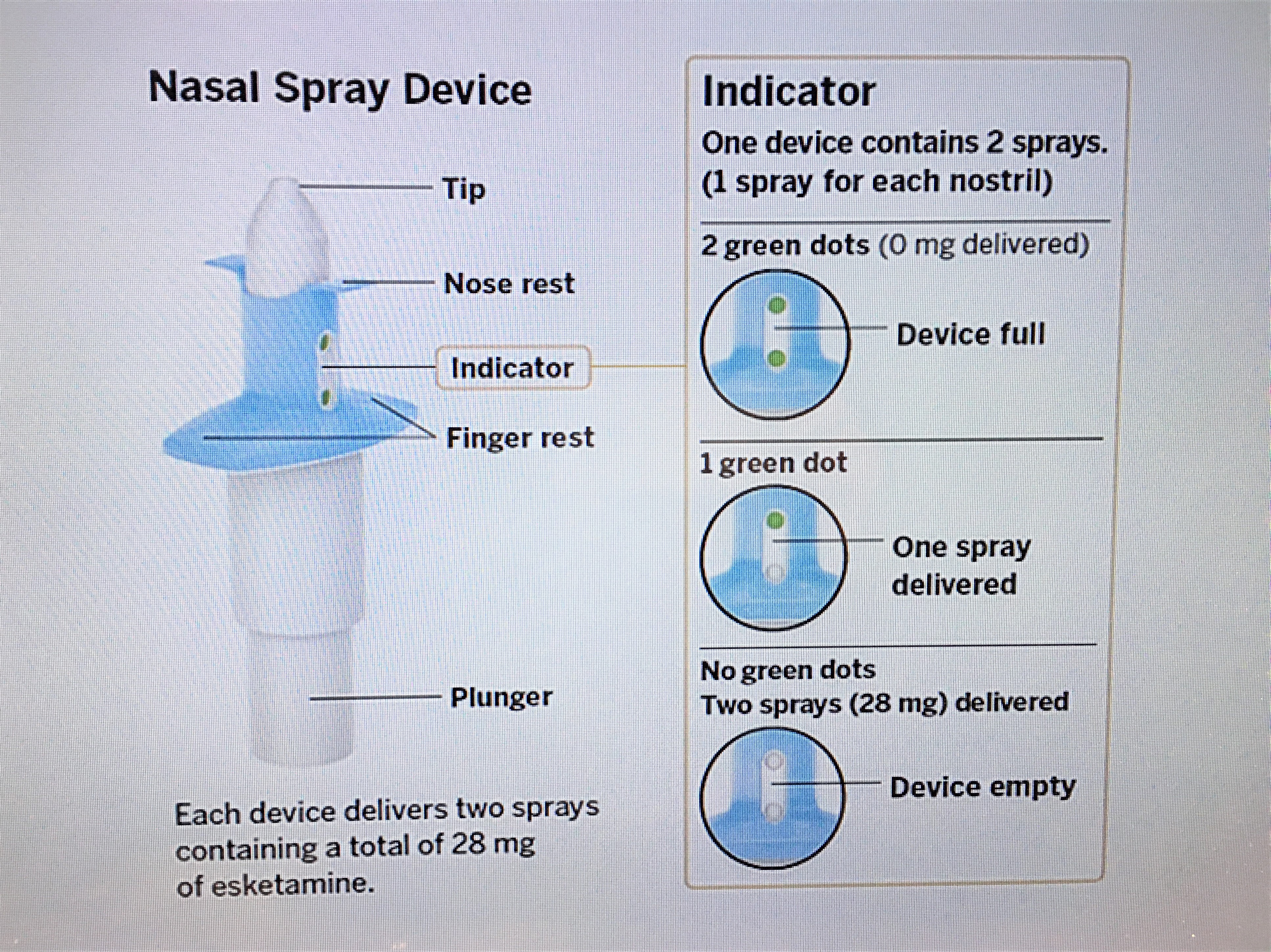
These highlights do not include all the information needed to use SPRAVATO® safely and effectively. See full prescribing information for SPRAVATO®. SPRAVATO® (esketamine) nasal spray, CIII Initial U.S. Approval: 1970 (ketamine)